EOS shares our vision to push the boundaries in medical
TCT MAG | May 2, 2022. Across the world, we have been left in no doubt in recent years about what can be achieved in the medical and healthcare sectors. Whether it is vaccination development and delivery programmes, advances in areas such as neuroscience or the remarkable engineering involved in creating artificial limbs, or other components for use in the body to improve quality of life.
Additive manufacturing (AM) has had a clear role to play in the healthcare sector and been pivotal to driving innovation in many areas. It is an area in which additive’s involvement is set to increase, with Grand View Research stating in a recent report that the healthcare AM sector could be worth $6.4bn by 2028.
A HERITAGE THAT COUNTS
The development of devices for use in the healthcare and medical sectors is rightly heavily regulated, whether a tool is used by a surgeon, a component for a hospital bed, or a device that will be used directly by a patient.
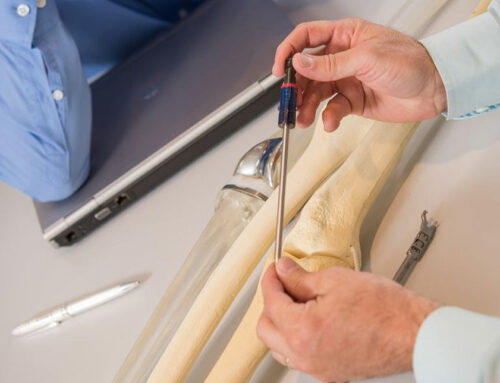
Precision, repeatability, reliability are all essential ingredients of producing high quality components. Additive manufacturing has shown itself to be more than capable of delivering on all these fronts, whilst also enabling some of the most revolutionary product designs, many of which could not be produced using traditional manufacturing methods. Working with metal, it can dramatically reduce the number of components required in an assembly, its weight, and the amount of handling required during production, which for medical applications directly lowers the risks of contamination. Add to this the ability to digitally customise parts such as plates and limb sockets to an individual patient’s needs, without retooling, and it is easy to see why the AM healthcare sector is predicted to grow so rapidly.

EOS M 300-4 additive manufacturing system: DMLS quality with up to 10 x more productivity (source: EOS)
EOS systems already produce over half a million parts a year and have been involved in the sector since 2006. EOS metal industrial 3D printers have an enviable heritage of use in heavily regulated industries, such as aerospace and medical series production. Recently, upgrades have enhanced their reputation for high quality and productivity even further, with many customers affectionately calling them a ‘true work horse’. Today, EOS has an install base of over 1,500 metal mid-frame systems, of which over 900 are EOS M 290 systems. Over half of these are qualified for production applications in regulated industries, such as medical.
Materials matter too when it comes to medical applications. Not only must they be suitable for placement in the body, but able to be precision printed into highly complex structures such as random lattice structures that allow the body’s natural processes to bond with an implanted component. One of the most common materials used in medical is Ti64ELI, which is excellent at meeting the reliability and repeatability requirements of medical use cases. An extensive capability study showed that EOS Titanium Ti64ELI coupled with an EOS M 290 has a minimum Four Sigma process capability for density, geometrical accuracy, and surface roughness properties: as well as Six Sigma capability for tensile and yield strength properties.
MAKING THE MOVE TO AM
There are few industries that innovate at the rate of the medical and healthcare sectors – it is a lifeblood – the desire to rid the world of a disease or improve the health and quality of life of individuals.
The journey from traditional manufacturing methods to successful AM is one that many EOS customers such as Lincotek in the healthcare sector have achieved with great success. At EOS, the Additive Minds consulting team helps customers take the leap into modern manufacturing through consulting, knowledge transfer and aiding the integration of industrial 3D printing into existing production settings.

Lincotek Additive Innovation Centre in Italy(Source: Lincotek)
A great product innovation idea is where every success story starts, but Additive Minds helps customers develop those ideas with a 3D printing mindset by exploring part design, materials, and manufacturing methods. Additive Minds also supports customers as they undergo qualification and certification for their designs – which, in the case of healthcare, demands some of the highest standards of any industry. Critically, Additive Minds advises on the wider plan to deliver products, helping customers not only ensure production is completely scalable, but ramps up in a way that is closely aligned with how the product will be introduced to different markets.
LINCOTEK ADDITIVE – A MEDICAL MARVEL
Lincotek Additive is part of global contract manufacturer Lincotek Group and has championed innovation in AM solutions since 2006. Lincotek is one of the pioneers in the AM space not only for medical implants, but other industries too. Lincotek started working with EOS early on, recognising that they offered one of the first powder-based processes able to support free-form printing with titanium alloys, and meet the standards required for implantable materials at a reasonable investment level.
Lincotek Additive has now grown to more than 25 EOS machines at locations in Italy, Switzerland, and the USA, using the EOSINT M 280, EOS M 290, EOS M 400-4 laser printers. In medical and orthopaedic applications alone, Lincotek produces over 100,000 implantable devices a year for use across the globe, including patient-specific prostheses from CT images. It is the biggest contract manufacturer in the space.
Lincotek Additive Innovation Centre enables24/7 AM industrial production and real-time monitoring of industrial 3D printers (Source: Lincotek)
EOS Additive Minds has worked closely with Lincotek throughout its expansion and growth to ensure consistent quality and efficient manufacturing processes across the production lifecycle at every stage. Currently, it is supporting Lincotek with achieving operational and process qualification for its new EOS M 300-4. A four-laser machine designed to deliver up to 5x productivity boost with consistent part quality across the entire 300x300mm build space, through its highly customisable hardware and software features. EOS and Lincotek are partnering to transfer specific additive solutions to the EOS M 300-4, as such ramping up a certified serial production on the latest AM technology available – for Lincotek’s and EOS customer’s future serial production needs.
Winfried Schaller, Lincotek, Group CEO, concludes: “Being a pioneer in the additive market is something that has allowed Lincotek to become the contract manufacturer of choice in the additive fi eld globally. Our focus is on serial production and end-to-end solutions in our ‘Additive Plus’ concept, allowing customers to have, for example, ‘ready to implant’ or ‘ready to engine’ parts from one source, reducing supply chain complexity and having a real alternative to traditional supply chains. As Lincotek Additive we off er our customers a real alternative in serial additive manufacturing, enhanced by profound knowledge in specific end-to-end capabilities such as heat treatment, hipping, coatings and precision machining. We are very excited to be in this fast growing sector offering unique, reliable and economic customer focused supply chain solutions, matching additive and traditional manufacturing technologies.
“Everything we do at Lincotek is about using the most innovate manufacturing techniques to deliver highest quality of parts for our customers. EOS shares our vision to keep the highest standards and push the boundaries of what can be achieved in the medical sector through AM techniques. Our continued growth and improved productivity are a testament to everyone involved in that process. To now cooperate again on the latest technology innovation with EOS on the M 300-4 is very exciting for us and will bring significant value to both existing and potential customers.”
Learn more about how EOS industrial 3D printers can support your manufacturing here.