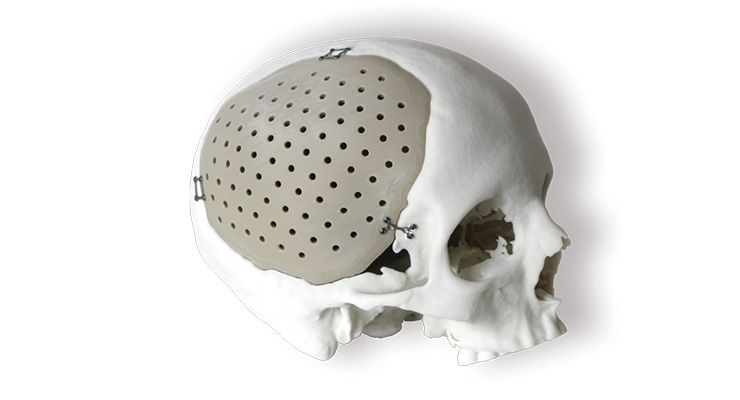
A customized, 3D-printed skull implant. Image courtesy of 3D Systems.
Eight experts in additive manufacturing for medical devices discuss the finer points of the promising sector.
In early October, Stryker released the latest addition to its portfolio of 3D-printed implants, the Monterey AL interbody system.
The anterior lumbar interbody fusion (ALIF) implant is 3D-printed via a proprietary process Stryker introduced years ago called AMagine additive implant manufacturing. The result is an implant composed of what Stryker calls Tritanium, a highly porous titanium material designed for bone in-growth biological fixation. Tritanium accomplishes this by mimicking cancellous bone, promoting bone regeneration and fusion.
“No one understands 3D-printing like Stryker—the fact that they’ve been able to dial in the right mix of small, medium, and large pores in a reproducibly randomized matrix is incredible,” Bala Giri, M.D., president and founder of the Texas Neuro Spine Institute, told the press.
Interbody fusion cages have been highly impacted by the additive manufacturing (AM) process, with many firms utilizing the manufacturing method to create porous structures that facilitate fusion. As a result of the burgeoning method, AM is much more in demand. This has led to a cavalcade of specialty manufacturers that employ AM among their services, and other firms entirely dedicated to the process.
The market has been exciting since its inception, and although it lagged behind somewhat for medical device manufacturing due to associated regulatory concerns, 3D printing has become an invaluable resource for device makers looking for a way to manufacture technologies with complex geometries.
To take a closer look at the AM/3D printing sector, MPO spoke with eight experts in the field:
- Justin Bushko, principal at Concise Engineering
- Cody Cochran, co-founder and general manager of Azoth
- Ben Johnson, VP, portfolio & regulatory, healthcare, 3D Systems
- Dan Owens, VP innovation and technology; and Dan Blum, VP/GM Pierceton Campus, Paragon Medical
- Greg Paulsen, director of application engineering at Xometry
- Francesco Robotti, technology and business development manager at Lincotek Medical
- Ron Zownir, manufacturing engineer at OKAY Industries
Sam Brusco: What latest additive manufacturing (AM) hardware or software advances have been most beneficial?
Justin Bushko: The one that has been most beneficial in medical device product development and prototyping has been the advancement of materials, primarily in plastics. We have been consistently building early-stage prototypes for the last five-plus years using SLA and resins, but they are always fragile and often degrade quickly or require substantial post-processing. The most recent advancements in FDM printing have provided actual plastics with significant strength, which also optimize the look and feel of the device. This, coupled with the advancements of the printers’ resolution to permit just a few microns per layer has improved the stepped features we’ve always battled. Additionally, the resins for SLA are now offering high-temperature and high-strength products for more accurate prints in stronger materials. We have even used some of the FDM and SLA prints to operate as molds for over-molding to improve turnaround of functional, appearance model prototypes. The new flexible materials in both SLA and FDM have significantly improved in the last couple of years. Some of the flexible materials are virtually indestructible by human strength, allowing for exceptionally durable prototypes for discussions on durometer, look and feel, and textures. This was not often possible a few years ago due to the resins degrading or remaining slightly uncured. These new advances in materials coupled with the hardware to adjust to the new materials and the software to reduce layer density increase support options and reduce post-processing have led to significant advancement in prototyping opportunities, allowing medical devices to be refined earlier in the design phase than possible just two to three years ago.
Cody Cochran: Sinter-based additive manufacturing, like binder jetting and material jetting, have been the most beneficial metal additive manufacturing technologies. These enable volumes previously not possible with additive manufacturing.
Ben Johnson: Recently, advancements in additive manufacturing capabilities have come to market from Kumovis, a 3D Systems company that opens new opportunities in polymeric implants from high-performance materials with clinical history such as polyether ether ketone (PEEK) and polyphenylsulfones like Radel (PPSU). The ability to print PEEK compliant to standard specifications for surgical implant applications has been difficult to achieve in additive manufacturing, but the solution is cost advantageous over traditional manufacturing and can be used to facilitate high mix, low volume product lines like what we see in personalized medical devices. The same is true with PPSU materials that can be leveraged for surgical instruments with the same capabilities as their surgical steel counterparts but at significant weight savings and a fraction of the price.
We continue to improve on our best-in-class manufacturing systems like the DMP Flex 350 metal 3D printer for fabricating parts in titanium, cobalt chrome, and stainless steel. The DMP Flex 350 Dual offers increased productivity and reduced operational costs for implant manufacturing but doesn’t sacrifice the high quality and advantageous powder management characteristics of our original systems. We have been working with customers to both qualify these systems for their current product lines, where we are seeing an up to 50% reduction in build times and also working to develop new products where better economics are needed, like in large joint replacement applications.
Innovations in software systems that bring together digital workflows and manufacturing operations are also helping to reduce costs and improve quality. We are making investments into software capabilities that automate workflows and reduce the potential for human error; both are targets that have a large implication for additive manufacturing costs and quality in healthcare. For instance, our Oqton software team works with customers to tailor manufacturing operating systems that help to automate part fixing, part labeling, print orientation, support generation, and part build parameters.
Dan Owens and Dan Blum: Paragon Medical is deploying the most up-to-date, state-of-the-art technology in both our Innovation Center and new 34,000-square-foot dedicated additive manufacturing site, including both 3D Systems DMP 350 and EOS printers. In addition, we utilize 3D Expert Software for the design process. That information is leveraged throughout our proposal generation, enabling a more informed design analysis and streamlining the transition to serial commercial production.
Greg Paulsen: AM is uniquely digital compared to traditional processes. Digital 3D files, build preparation software, and cloud-based manufacturing execution systems help create repeatable and auditable 3D prints. The latest advancements in additive manufacturing are software-enabled tools that create digital twins of the product. These digital twins can provide a predicted outcome and adjust the machine input in the most advanced products to make prints more reliable and accurate. Furthering this are tools to monitor and adjust build settings during the 3D printing process, reviewing the results layer by layer and comparing reality against simulation to improve quality control.
This symbiosis of software and hardware is necessary for medtech industries to develop and produce parts using 3D printing with the same confidence as molded or machined counterparts.
Francesco Robotti: In the software, there have been advancements in developing algorithms to help the segmentation phase, accelerating the creation of 3D models starting from diagnostic images. Software was also developed to help design complex shapes to accommodate personalized anatomy. Other software is helping design of porous-lattice structure, simplifying the computational work for the benefits of faster design and faster printing execution.
For hardware, multiple power sources in powder bed fusion (PBF) are helping to achieve faster completion of heavy jobs, e.g., when a big number of units have to be delivered with rolling cadence. On the other hand, small equipment has shown to be flexible and efficient when small items or few units are the typical load for manufacturing requested.
Ron Zownir: Rapidia’s 3D metal printing system is open, versatile, and economical. It includes a printer with two independent print heads fed with Rapidia’s water-based metal and support material pastes, as well as a sintering furnace where parts can be sintered overnight instead of taking days.
Brusco: Which medical sectors and/or manufacturing process steps is AM benefitting most prominently?
Bushko: While titanium and ceramic 3D printing has advanced to use in medical device finished goods, plastics have still not crossed that threshold as quickly. There are many challenges when it comes to durability and rigidity of the materials with respect to usage of the tools in a surgical instrument. Additionally, the primary guidance of the FDA forces the validation of the process even further than injection molding, with many not yet equipped to handle for plastics. Additionally, there are many challenges using an FDM printed component to mount circuit card assemblies due to the electrical product safety requirements. I expect there will be more use of simple, non-safety components, such as spacers, or other features to provide isolated compartments where true electrical isolation is not a factor. For example, a cart could have the tube caps 3D printed, but you would not likely use a 3D printer mounting bracket for the isolation transformer. The impacted titanium products are currently benefitting the industry most directly, allowing ingrowth and other more complex geometries, which would be nearly impossible with machining. Additionally, the AI aspects of the software to reduce weight and overall structure while ensuring strength post-validation provides unbelievable options. The early stage of the design also significantly benefits the industry, but this just helps cost, speed, and commitment early in the feasibility and design phases. The consumer never truly sees those benefits unless it results in speed to market or reduction in overall cost of components that is passed on. Otherwise, these benefits are simply realized in the product’s development costs, saving the design owner time and money before advancing to injection molding.
Cochran: AM is significantly benefiting the medical surgical tooling space. AM allows rapid product development and sinter-based additive manufacturing can even enable low-mid volume production before metal injection molding starts to make sense.
Johnson: Additive manufacturing has over two decades of history as a technology used to augment patient care in various specialties. Most prominently these solutions have been deployed where customization provides a significant benefit to the clinician or patient like in dentistry, craniomaxillofacial surgery, and prosthetics. Additionally, serial production of orthopedic and spine devices has been a great success story where better economics can be achieved in the manufacture of devices with features not easily obtained from traditional methods. More recently, applications in medical specialties such as radiation oncology and pharmaceuticals have also shown promise. I think it would be difficult to find a medical device company that doesn’t leverage additive manufacturing at some point in their process, from prototyping to testing to device production.
Owens and Blum: AM allows design and manufacture of concepts that would not have been possible to create historically with subtractive manufacturing, including specifically designed surface features and finishes to promote biologic fixation. Complexity is reduced with AM, as it offers a more dynamic design-driven process. We have been working with several OEM clients on both implant and instrument designs that take advantage of the ability to design not only the final shape but also surface texture and bone ingrowth surfaces. Most of our clients today are in the orthopedic market space, one of the most dynamic segments—with AM growth estimated at almost 20% per year. In addition to providing our clients with expertise in AM, we deliver operational excellence in AM post-processing as well as end-to-end supply chain management.
Paulsen: Although direct pharmaceutical or biotech 3D printing is the future, the current state of 3D printing is best for mechanical goods. Rapid components’ production without the need for upfront tooling makes 3D printing prominent in medical device product development and custom tooling used in pharmaceutical production. Industrial AM options continue to expand in material selection, and surface finishing, meeting more customer needs further up the product development stream. There are a few instances where direct 3D manufacturing of medical goods has played a role, such as nasal swabs, but these efforts require significant investment and validation.
Robotti: Lincotek’s Medical Division operates in the orthopedic market only; from this limited perspective, a lot of subsegments were heavily impacted. For example, AM is fully used to deliver implantable Ti 6-4 parts. This is quite a niche! In some applications like intervertebral spacers, this combination already covers about one-third of the market, and this share is still growing, headed to become in the future the most used technology. When we move our attention to the joint implants, the use of Ti6-4 and AM is still a fraction of market. However, it showed clinical benefit in the hip, shoulder, and knee, the three most important implant segments by annual total volumes. If we move out from serial production for custom-made implants, 3D printing is already the leading technology to deliver individual solutions in a short time. Non-implantable polymer materials and AM are the elective choice for a lot of surgical ancillary devices like bone and implant replicas, cutting jigs, etc. These 3D-printed devices help surgeons in planning and executing their surgery.
Zownir: Reducing toolroom burden is an important benefit of AM. Metal AM can be used to create tooling and fixtures and allows toolmakers to focus on work where their precision skills are most essential to success. Enhancing prototyping service is another benefit of AM. OKAY’s NexTech Labs R&D labs with fully dedicated resources facilitate early customer collaboration on product development and assist with design for manufacturing and product launch. 3D metal printing working in tandem with CNC machining can reduce lead time, allowing customers to iterate more quickly.
Brusco: Please describe a noteworthy AM project. Why was it noteworthy?
Bushko: In the development of surgical tooling, we have been able to advance our prototyping from day one sketching and industrial design to semi-functional prototype by day 30. This has been a substantial change from years past, requiring fully defined 3D models to be developed to enable machining of plastics, which often took two to three months, depending on the resources available. Additionally, any minor errors or client recommendations within this path could add weeks or months to the critical path. Additionally, these machined components could cost more than an inexpensive 3D printer these days. In concrete terms, we have been able to reduce our time from sketch to prototype by 70-80% and reduce the component costs by 70-80% as well. These wins are significant in early-stage product development. Additionally, the look and feel of the prototypes these days as opposed to just two to three years ago is easily 4-5x improved over prior capabilities. Having successfully completed multiple sketches to semi-functional prototypes in under 60 days has proven extremely useful in recent design projects.
Cochran: Azoth has helped companies working in the robotic surgery space decrease their time to market by utilizing sinter based additive manufacturing to shorten product development timelines. Our small complex metal part solutions help customers rapidly iterate designs and enable low-volume production on the exact same equipment. Rather than utilizing 3D printing, then machining, and finally MIM (metal injection molding), we are able to use the exact same platform. This eliminates the need for designing products to be manufactured multiple different ways and allows customers to utilize “near-production-ready” components within 10 days of ordering components.
Johnson: There are a few noteworthy projects that we’ve been working on recently. First, we think there is an opportunity in radiotherapy to augment patient care with better methods to fabricate the accessories used to ensure proper dosage delivery. We’ve started on this endeavor with a product we recently received FDA clearance for called VSP Bolus, an accessory used to control the depth of radiation received by the patient.
Second, an endeavor of collaboration we started with the Veterans Administration to develop medical device manufacturing capabilities at the point of care has resulted in several applications being deployed to improve veteran care. Most recently, this collaboration resulted in the VA receiving FDA clearance for a craniofacial surgical planning and instrumentation product. This marks a major milestone in what could be a transformative factor in the medical device industry, with a new look at how hospital systems can control their supply chains and develop products to serve unmet clinical needs.
Another exciting announcement we just made in a field ripe for innovation with additive manufacturing is the formation of a wholly owned company focused on drug discovery and development called Systemic Bio. This company will leverage our ongoing work in regenerative medicine to provide solutions to pharmaceutical companies looking to accelerate and reduce costs in drug testing. We think using additive technologies including materials developed specifically for regenerative medicine can dramatically reduce the reliance on animal testing.
Owens and Blum: We recently partnered with an OEM, strategic additive manufacturer, and an orthopedic startup business to bring a novel large joint implant design to market. Through our expertise in AM DFM at our Innovation Center, manufacturing, and post-processing, the team has enabled commercial success for our OEM client while driving efficiencies and streamlining their supply chain through end-to-end integration. This novel implant design is the first of its kind in the large joint orthopedic market and is manufactured without the use of forging or casting tooling. Due to our ability to quickly modify concept designs and 3D print prototypes for this client, the team was able to provide design iterations and improvements within hours to be tested at our onsite Bioskills lab with the OEM’s surgeon design team. AM has provided this client with a faster time to market by reducing the operational steps required to manufacture this implant, resulting in their commercial success.
Paulsen: 3D printing in metals and plastics has become a standard for custom orthopedic devices, prosthetics, and other assistive technologies. Clients utilize Xometry’s extensive AM offerings to produce these devices for items such as custom orthotic inserts, knee braces, prosthetic socket models, and assistive tools. Typically, these items would require expensive and time-consuming casting or molding, and additive alternatives have become a novel solution for iterative prototyping and end-use medical devices due to the cost and competitiveness.
Robotti: We have been involved in manufacturing individualized implants. This is a type of challenge that impacts the entire manufacturing structure of a company. Efficiency is a high hurdle to leap when you must dedicate a full manufacturing line, adjust your quality system and quality controls, stretching your validations to include these unique devices. Delivery time and design-to-part in the OR, needs to respect before unmatched tight calendar cadences, with very limited margin for adjustment.
In this context, manufacturing at the point of care could become a disruptive business model. Complexity and criticality of parts, including implants printed at the hospital, are rapidly growing. Even if a pilot project is considered far-fetched, it has the value to enlighten anyone in some key center of excellence that it’s possible to deliver data instead of items to solve a specific clinical issue. That’s a great concept, in my opinion. However, regulatory is still behind the possibility opened by the technology.
Zownir: 3D metal printing allowed us to enhance design, test, and produce a production proven prototype fixturing part within two days.
Brusco: What barriers to wider medical AM adoption still exist, and how might they be overcome?
Bushko: In medical device product development, the primary concerns for materials: material compatibility, biocompatibility, reliability, durability, accuracy, and potential flaws. As the hardware, software, and materials are now able to create a print more consistently, there will be more focus on the validation of these components. The type of failures possible with layers delaminating, voids, support post-clean up variation, remaining volatile chemicals due to lack of curing, and other issues will continue to need to improve and reduce the impact of these challenges on the final production component. Some of these issues are being resolved by the metal and ceramic service bureaus as they review the final chemical analysis and inspect for structure, voids, remaining powder, or binding materials, etc. These practices will eventually become more consistently accepted and drive the outcome of the print to meet these expectations. As this continues, the AM industry will need to adapt to these expectations. As long as the industry continues to improve, and the scrap ratio continues to beat typical machining and injection molding timelines and costs, the AM industry will continue to advance and more AM components will become adopted as production components.
Cochran: Pioneers in the AM space who focus on quality and adhering to traditional manufacturing standards make great strides toward making this a more widely adopted production method.
Johnson: The barriers to adoption are similar to what we’ve seen and worked on over the last few years in terms of regulations, clinical data, and costs. Solving some of these hurdles will lead to wider adoption, but despite these challenges, we see positive momentum in the industry. For example, large facility investments are being made by several companies to increase additive manufacturing capacities. Another signal we see is the incremental improvements customers are making to existing product lines that use additive manufacturing. The market is telling us that AM is primed for increased adoption, but in regulated industries, this will take time.
One way we are answering the call for increased awareness and adoption is by participating with regulators and standards committees to set recognized consensus documents that provide information and know-how for additive manufacturing. This will help to ensure safety and reliability across the industry.
Clinical data, especially data related to personalization of implants, need to be studied with more depth and power than what is currently available. Level 1 clinical evidence is needed to understand the costs and benefits of patient-specific applications, and these datasets will help to set the table for increased adoption through provider acceptance and payor reimbursements. I anticipate over the next several years we will start to see more compelling Level 1 studies published, and with that evidence, increased medical adoption of additive manufacturing solutions.
Owens and Blum: AM has been a viable manufacturing method for several years. However, the need to streamline and minimize complexity for our clients’ supply chains continues to be a need. Through our expertise in not only AM but also AM post-processing and supply chain management, we are able to offer a full end-to-end solution to our clients. Market acceptance of AM will continue to grow through education of the technology and partnership of OEMs with unique CMOs that offer capabilities backed by R&D and operational expertise.
Paulsen: Like injection molding or machining, AM acts as a production tool with its strengths and trade-offs. 3D printing remains supreme in low-volume, high-mix projects such as prototyping, but requires design and post-processing efforts to match end-use production. The ways that additive manufacturing could overcome these barriers are with software advancements, materials expansion and validation, and novel means of post-processing to seal surfaces.
Robotti: There are technical features yet to reach the range of applications in orthopedics, for example delivering increased fatigue-resistant bulk metals for load-bearing applications. This needs a combination of efficient manufacturing approach and efficient quality controls to stay cost competitive. Another example is to get items natively ready for use without post-processing like smoothing, thermal treatments, and the need for final machining assembling features.
There are also regulatory barriers that require serious investments to collect all the evidence and the inherently long time to market associated with this issue. These are the rules of the game but we can properly design for additive manufacturing to run a successful business case. We need to find ways to position our AM items out of the crowd because of their unique features. Each initiative must have some clear value proposition to catch a distinguished price premium, retain it for a long while, and pay back the effort.
Zownir: Currently most companies design for conventional manufacturing. Breaking through in adopting a design for additive manufacturing process requires experience, comfort, and confidence in metal additive part making. Designing for AM and playing to the strengths of the technology maximizes its impact, yielding parts that can be lighter, simpler to assemble, higher performing, and easier or more economical to produce.