PLASMA SPRAY COATING ON PEEK
Our long-time experience in treating metallic implants with air plasma spray and vacuum plasma spray technologies was used as the basis for the development of a new plasma spray coating family for polyetheretherketone (PEEK)-based components.
The family includes Spondycoat® and Ti-Growth®, which are highly adherent to PEEK and offer enhanced osseointegration when compared to uncoated PEEK devices.
Why coat PEEK?
PEEK is a biocompatible, inert, biomaterial that has been used to make orthopedic and spinal implants and trauma components for decades. Some concerns have been raised about its inertness and its limited fixation to bone.
Using our plasma spray technology and know-how to effectively coat PEEK devices with osteoconductive materials such as hydroxyapatite (HA) or titanium (Ti), enables a direct osseointegration to be achieved.
Medical devices made of PEEK that can be coated include interbody fusion devices, vertebral body components, artificial discs, and fixation screws.
Coatings for PEEK
Our current family of plasma spray coatings for PEEK includes:
- Spondycoat®-T 371A
- Spondycoat®-T 107
- Ti-Growth®
- Spondycoat®-HA
All of these coatings can be applied to devices made of plain PEEK, PEEK reinforced with short carbon fiber, and barium sulfate-filled PEEK. Further coating solutions are available or may be developed upon specific request.
All Spondycoat® and Ti-Growth® coatings have characteristics in compliance with ASTM standards and FDA guidance.
A comprehensive data set about the influence of the plasma spray process on PEEK performances has been collected and is available upon request.
This information may help device designers in choosing the proper coating for the specific application.
Contact us
for more details or download our brochure which includes information about the biological characterizations of our plasma coatings for PEEK.
SPONDYCOAT®-T 371A TI COATING
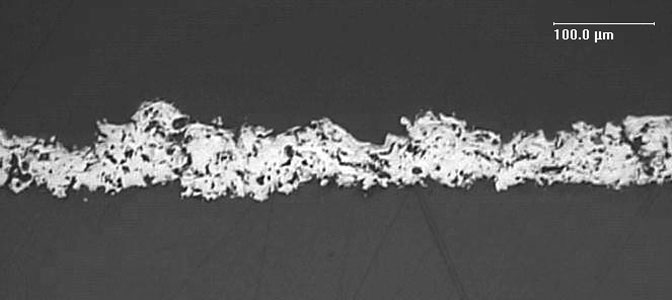
Spondycoat® T371A is a Ti coating with low roughness (representative values of Ra 4–10 microns) mainly indicated for thin layers (recommended thickness 60–120 microns) on medical devices.
SPONDYCOAT®-T 107 TI COATING

Spondycoat®-T 107 is a Ti coating with high roughness (representative values of Ra 20-40 microns) and a recommended thickness of 125–250 microns.
Ti-Growth® TI COATING

Ti-Growth® is a porous Ti coating with high roughness (representative values of Ra 40–80 microns and a recommended thickness of 300–500 microns.
Spondycoat®-HA HA COATING

Spondycoat®-HA is an HA coating with low roughness (representative values of Ra 4-8 microns) mainly indicated for thin layers (recommended thickness 45–85 microns).

